
cobas b 123 POC system
Instructions for Use, Version 13.0
Software version 4.17

cobas b 123 POC system
Roche Diagnostics
2 Instructions for Use · Version 13.0
Publication information
Revision history
Edition notice This publication is intended for operators of the cobas b 123 POC system.
Every effort has been made to ensure that all information contained in this
publication is correct at the time of publishing. However, Roche may need to update
the publication information as output of product surveillance activities, leading to a
new version of this publication.
General attention
To avoid serious or fatal injury, ensure that you are familiar with the system and safety
information before you use the system.
r Pay particular attention to all safety precautions.
r Always follow the instructions in this publication.
r Do not use the instrument in a way that is not described in this publication.
r Store all publications in a safe and easily retrievable place.
Training Do not carry out operation tasks or maintenance actions unless you or your trainer
have received training from a Roche representative. Leave tasks that are not described
in the user documentation to trained Roche Service representatives.
Screenshots The screenshots in this publication have been added exclusively for illustration
purposes. Configurable and variable data, such as tests, results, or path names visible
therein must not be used for laboratory purposes.
Manual Version Software Version Revision date Changes
1.0 November 2009 not delivered
2.0 2.0 November 2010 Launch
2.1 April 2011 FDA submission draft
3.0 2.3 August 2011
4.0 2.4 December 2011
5.0 3.0 July 2012
6.0 4.0 December 2012 internal release - not launched
7.0 4.0 January 2013
8.0 4.1 July 2013
9.0 4.5 April 2014
10.0 4.7 February 2015
10.1 4.8 September 2016 Software update
11.0 4.13 October 2017 Software update
12.0 4.15 February 2019
o Safety messages update
o Data protection (GDPR)
update
o Software updates
13.0 4.17 August 2020
o Hardware update
o Safety message updates
o Added hypodermic needle as
consumable
Table 1 Revision history

Roche Diagnostics
Instructions for Use · Version 13.0 3
cobas b 123 POC system
Warrant y Any customer modification to the system renders the warranty or service agreement
null and void.
For conditions of warranty, contact your local sales representative or refer to your
warranty contract partner.
Copyright © 2009-2020, F. Hoffmann-La Roche Ltd. All rights reserved.
License information cobas b 123 POC system software is protected by contract, copyright law and
international treaties. cobas b 123 POC system contains a user license between F.
Hoffmann-La Roche Ltd. and a license holder, and only authorized users may access
the software and use it. Unauthorized use and distribution may result in civil and
criminal penalties.
Open Source and Commercial
Software
cobas b 123 POC system may include components or modules of commercial or
open-source software. For further information on the intellectual property and other
warnings, as well as licenses pertaining to the software programs included in the
cobas b 123 POC system, refer to the electronic distribution included with this
product.
This open source and commercial software and the cobas b 123 POC system as a
whole can constitute a device regulated in accordance with applicable law. For more
detailed information, refer to the Instructions for Use and labeling.
Please note that the respective authorization is no longer valid according to the
corresponding legislation should any unauthorized changes be made to the
cobas b 123 POC system.
Trademarks The following trademarks are acknowledged: COBAS, COBAS B, LIFE NEEDS
ANSWERS, AUTOQC, ROCHE MICROSAMPLER and COMBITROL are
trademarks of Roche.
All other trademarks are the property of their respective owners.
Feedback Every effort has been made to ensure that this publication fulfills the intended use. All
feedback on any aspect of this publication is welcome and is considered during
updates. Contact your Roche representative, should you have any such feedback.
Approvals The cobas b 123 POC system meets the requirements laid down in:
Directive 98/79/EC of the European Parliament and of the Council of 27 October
1998 on in vitro diagnostic medical devices.
Directive 2011/65/EU of the European Parliament and of the Council of 8 June 2011
on the restriction of the use of certain hazardous substances in electrical and
electronic equipment.
This instrument complies with the emission and immunity requirements described
in this part of the IEC 61326 series.
Compliance is provided by means of the Declaration of Conformity.
The following marks demonstrate compliance:
For in vitro diagnostic use.
Complies with the provisions of the applicable EU directives.

cobas b 123 POC system
Roche Diagnostics
4 Instructions for Use · Version 13.0
Contact addresses
Manufacturer
Edition
Version 13.0, August 2020
First edition: November 2009
Issued by TÜV SÜD for Canada and the US.
Laboratory Equipment is the product identifier as shown on the
name plate.
Roche Diagnostics GmbH
Sandhofer Strasse 116
68305 Mannheim
Germany
Made in Switzerland
www.roche.com

Roche Diagnostics
Instructions for Use · Version 13.0 5
cobas b 123 POC system
Table of contents
Publication information 2
Contact addresses 4
Edition 4
Table of contents 5
Intended use 7
Intended user 7
Use environment 7
Symbols and abbreviations 7
What is new in this version 12
Introduction and specifications
1 Safety information
Safety classifications 17
Safety precautions 18
Safety labels on the instrument and
consumables 26
2 General description
System information 29
System components 31
Consumable information 41
3 Specifications
Performance data 55
Sample throughput 79
Sample measurement time 80
Sample volumes 81
Sample types 82
Calibrations 83
Environmental parameters 84
Product data 86
Printer 88
User interface module 89
Barcode scanner 90
Imaging scanner (optional) 92
4 Theoretical foundations
Assigning unit formats to parameters 95
Selecting between pH and H
+
unit formats 96
Parameters and calculations 97
Calibration principle 113
Oximeter module (optional) 114
Reference and critical values 115
Assigning reference and critical ranges 118
Monitoring reference and critical ranges on
measurement reports 121
Assigning a correlation factor to a parameter 122
Operation
5 Installation and put out of operation
Installation 129
Installing the instrument 131
Put out of operation 134
6 Measurement
Preanalytics 139
Interferences 145
Limitations of clinical analysis 152
Configuring measurement input values 154
Configuring the measurement results screen 156
Configuring the parameter selection field 157
Performing a measurement 160
Measurement modes 169
Entering measurement input values 170
Measurement results and reports 173
Measuring database 175
Acid base maps 176
Patient trend diagrams 177
7 Quality control
General QC concept 181
Important information for evaluating QC
results 183
Performing QC measurements 184
QC results and reports 191
QC database 192
Levey-Jennings graphs 193
Performing a proficiency test measurement 194
Configuring the QC scheduler 199
Defining and configuring QC materials 203
Configuring QC rules and consequences 207
Configuring QC settings 209
Evaluating QC measurement results 211
Multirules 212
Removing QC consequences 215
QC troubleshooting 218
Troubleshooting AutoQC module blockages 219
8 Calibration
General information 223
Automatic calibrations 225
User-activated calibrations 228
Calibration database 229
Configuring calibration settings 230

Roche Diagnostics
6 Instructions for Use · Version 13.0
cobas b 123 POC system
9 Calibration verification control
General information 235
Installing AutoCVC Packs 236
Performing an AutoCVC measurement 238
Managing CVC results in the QC database 240
Configuring analytical measurement ranges 241
10 Software functions
General software information 247
Overview tab 251
Description of the screen areas 252
Workplace tab 255
Instrument tab 256
Utilities tab 257
Instrument databases 258
Logging on and off 260
Connecting USB storage devices 261
Configuring system settings 262
Configuring network connection settings 269
Configuring operator settings 278
Changing security levels 282
Performing software and language updates 284
Exporting and importing configuration
settings 285
Audit trail 286
Available printout reports 288
Consumable change
11 Consumable change
Disinfection procedures 299
Disinfecting the instrument 301
Changing consumables 303
Additional information for consumable
changes 307
Troubleshooting
12 Troubleshooting
General information 313
Alarm area and buttons 314
System stops 316
Defects 320
Warnings 321
Information alarms 322
Status messages on the measurement report 323
Calibration and QC troubleshooting for
parameters 324
Performing Sensor Cartridge wetting routines 326
Checking the Fluid Pack valve positions 327
Barcode scanner troubleshooting 328
Creating troubleshooting reports and log files 329
Depot repair 330
Appendix
13 Available accessories and consumables
Available accessories and consumables 335
14 Copyright information
Software license information 341
15 Glossary
Index
Index 349

Roche Diagnostics
Instructions for Use · Version 13.0 7
cobas b 123 POC system
Intended use
The cobas b 123 POC system is a fully automated POC system for in vitro
measurement of pH, blood gases (BG), electrolytes (ISE), hematocrit (Hct),
metabolites (Glu, Lac), total hemoglobin (tHb), hemoglobin derivatives (O
2
Hb,
HHb, COHb, MetHb), oxygen saturation (SO
2
) and neonatal bilirubin (Bili).
In addition, the cobas b 123 POC system calculates derived parameters.
It is dedicated for use in a Point-of-Care environment and laboratory.
The integrated AutoQC module and the oximeter module are available as an option.
Intended user
Users of the cobas b 123 POC system are trained professionals in the laboratory and
trained healthcare professionals in a point-of-care environment.
Use environment
The cobas b 123 POC system is intended to be operated in point-of-care settings and
hospital laboratories.
Symbols and abbreviations
Product names Except where the context clearly indicates otherwise, the following product names
and abbreviations are used.
Symbols used in the publication
Product name Descriptor
cobas b 123 POC system system
cobas b 123 Fluid Pack Fluid Pack
cobas b 123 Sensor Cartridge Sensor Cartridge
cobas b 123 AutoQC Pack AutoQC Pack
cobas b 123 AutoCVC Pack AutoCVC Pack
cobas b 123 Printer Paper printer paper
Table 2 Product names
Symbol Explanation
o List item
r Start of a task
s End of a task
u Related topics containing further information
q Tip. Extra information on correct use or useful hints.
Table 3 Symbols used in the publication

cobas b 123 POC system
Roche Diagnostics
8 Instructions for Use · Version 13.0
Symbols used on the instrument
Symbols used on consumables
Symbol Description
Catalog number
Serial number
Global Trade Item Number
Manufacturer
Date of manufacture
Direct current
Caution
Biohazard
Table 4 Symbols used on the instrument
Symbol Description
Catalog number
Batch code
Global Trade Item Number
Use by date
Temperature limitation
Table 5 Symbols used on consumables

Roche Diagnostics
Instructions for Use · Version 13.0 9
cobas b 123 POC system
Do not use if package is damaged
Consumable contents
Control material
Fragile handle with care
Store upright
Do not reuse
Manufacturer
Sterilized using irradiation
Sterilized using ethylene oxide
Sufficient for <n> tests
Operator’s manual/operating instructions
Caution, consult accompanying documents
Handle with care
Software symbol for the Fluid Pack
Symbol Description
Table 5 Symbols used on consumables

cobas b 123 POC system
Roche Diagnostics
10 Instructions for Use · Version 13.0
Abbreviations
Software symbol for the AutoQC Pack
Software symbol for the Sensor Cartridge
Biohazard
Symbol Description
Table 5 Symbols used on consumables
Abbreviation Definition
1P 1-point calibration
2P 2-point calibration
AMR Analytical measurement range
A
rate
Assisted breathing rate
ASTM American Society for Testing and Material
AutoQC Automatic Quality Control
BG Blood gas
Bili Bilirubin
BSA Bovine serum albumin
Ca
2+
Ionized calcium
CCD Charge coupled device
CE Conformité Européenne
CF Compact flash
Cl
-
Chloride
CLIA Clinical Laboratory Improvement Amendments
CLSI Clinical and Laboratory Standards Institute
COHb Carboxyhemoglobin
COOX Co-oximetry
CPAP Continuous positive airway pressure
CSV Comma-separated values
CVC Calibration verification control
DHCP Dynamic host configuration protocol
DIN German national organization for standardization
DNS Domain name server
e.g. for example
EC European community
EN European standard
EQA External Quality Assurance
Glu Glucose
H
+
Hydrogen ion concentration
Hb Hemoglobin
Hct Hematocrit
Table 6 Abbreviations

Roche Diagnostics
Instructions for Use · Version 13.0 11
cobas b 123 POC system
HHb Deoxyhemoglobin
i.e. that is to say
IEC International Electrotechnical Commission
ISE Ion-selective electrode
ISO International Organization for Standardization
IT Information technology
IVD In vitro diagnostic
K
+
Potassium
Lac Lactate
LCD Liquid crystal display
LED Light-emitting diode
LIS Laboratory Information System
MAP Mean airway pressure
MCHC Mean corpuscular hemoglobin concentration
MCT Medium chain triglycerides
MetHb Methemoglobin
mm millimeter
MSDS Material safety data sheet
MV Mean value
Na
+
Sodium
NIST National Institute of Standards and Technology
O
2
Hb Oxyhemoglobin
PCO
2
Partial pressure of carbon dioxide
PEEP Positive end-expiratory pressure
PIP Peak inspiratory pressure
PO
2
Partial pressure of oxygen
POC Point-of-care
POCT1-A Connectivity standard for point of care devices
QC Quality control
RAM Random access memory
RECAL Recalibration
RQ Respiratory quotient
SD Standard deviation
SD card Secure digital memory card
SIM Sample input module
SO
2
Oxygen saturation
S
rate
Spontaneous breathing rate
STDBY Standby
Te Expiratory time
tHb Total hemoglobin
Ti Inspiratory time
Abbreviation Definition
Table 6 Abbreviations

cobas b 123 POC system
Roche Diagnostics
12 Instructions for Use · Version 13.0
What is new in this version
o Updates for new hardware version
o Updated safety messages
o Added Hypodermic needle as consumable
o Editorial updates.
UIM User interface module
USB Universal Serial Bus
VT Tidal volume
Abbreviation Definition
Table 6 Abbreviations

Introduction and specifications
1 Safety information ........................................................................................................................... 15
2 General description.......................................................................................................................... 27
3 Specifications .................................................................................................................................... 51
4 Theoretical foundations ................................................................................................................... 93


Roche Diagnostics
Instructions for Use · Version 13.0 15
cobas b 123 POC system 1 Safety information
Table of contents
Safety information 1
The chapter provides information about the safe operation of the system.
In this chapter
Chapter
1
Safety classifications.............................................................................................................17
Safety precautions.................................................................................................................18
Operator qualification...................................................................................................18
Safe and proper use of the system................................................................................18
Installation and deinstallation......................................................................................19
Operating conditions.....................................................................................................19
Electrical safety...............................................................................................................20
Biohazardous materials .................................................................................................20
Mechanical safety...........................................................................................................21
Consumables...................................................................................................................21
Reagents and working solutions...................................................................................22
Incorrect results..............................................................................................................23
Software and data security............................................................................................24
Instrument damage........................................................................................................25
Safety labels on the instrument and consumables ...........................................................26

Roche Diagnostics
16 Instructions for Use · Version 13.0
1 Safety information cobas b 123 POC system
Table of contents

Roche Diagnostics
Instructions for Use · Version 13.0 17
cobas b 123 POC system 1 Safety information
Safety classifications
Safety classifications
This section explains how precautionary information is presented in this manual.
The safety precautions and important user notes are classified according to the ANSI
Z535.6 Standard. Familiarize yourself with the following meanings and icons:
Safety alert
The safety alert symbol is used to alert you to potential physical injury hazards. Obey all
safety messages that follow this symbol to avoid possible damage to the system, injury, or
death.
These symbols and signal words are used for specific hazards:
WARNING
WARNING
r ... indicates a hazardous situation that, if not avoided, could result in death or serious
injury.
CAUTION
CAUTION
r ... indicates a hazardous situation that, if not avoided, could result in minor or moderate
injury.
NOTICE
NOTICE
r ... indicates a hazardous situation that, if not avoided, may result in damage to the
instrument.
Protective gloves and goggles, suitable protective clothing, and if necessary, mouth
protection has to be worn.
Important information that is not safety relevant is indicated with the following
symbol:
q
Tip
... indicates additional information on correct use or useful tips.

Roche Diagnostics
18 Instructions for Use · Version 13.0
1 Safety information cobas b 123 POC system
Safety precautions
Safety precautions
CAUTION
To avoid serious or fatal injury, read and comply with the following safety
precautions.
Operator qualification
Insufficient knowledge and skills As an operator, ensure that you know the relevant safety precaution guidelines and
standards and the information and procedures contained in these instructions.
o Do not carry out operation and maintenance unless a Roche representative has
trained you or your trainer to do so.
o Leave maintenance, installation, or service that is not described to trained Roche
Service representatives.
o Carefully follow the procedures specified in the instructions for operation and
maintenance.
o Follow standard laboratory practices, especially when you work with
biohazardous material.
Safe and proper use of the system
WARNING
Personal injury and infection due to sharps, rough edges, and/or moving parts
r Good Laboratory Practice can reduce the risk of injury. Be aware of your laboratory
environment, well-prepared, and follow the instructions for use. Some areas of the
instrument may have sharps, rough edges, and/or moving parts. Wear personal
protective equipment to minimize the risk of injury from bodily contact with such parts,
especially in less accessible areas, or while cleaning the instrument. Your personal
protective equipment should be appropriate to the degree and type of potential hazard,
e.g. suitable lab gloves, eye protection, lab coat, and footwear.
WARNING
Missing personal protective equipment
Working without personal protective equipment means danger to life or health. Using the
instrument in a way that is not described by the manufacturer can impair the protection
provided.
r Wear appropriate personal protective equipment, such as:
o Lab gloves
o Lab coat
o Eye protection
o Face shield if there is a chance of splashing or splattering
System not used for an extended
period
o If the system will not be used for more than 24 hours, put the system out of
operation.
o Remove and refrigerate any remaining reagents.
o For further information, call your Roche Service representative.
Non-approved parts Use of non-approved parts or devices may result in malfunction of the system and
may render the warranty null and void.
o Use only parts and devices approved by Roche Diagnostics.

Roche Diagnostics
Instructions for Use · Version 13.0 19
cobas b 123 POC system 1 Safety information
Safety precautions
Installation and deinstallation
Transport Risk of personal injury when lifting. The cobas b 123 POC system, without
consumables in the bottle compartment and AutoQC materials in the AutoQC
module, weighs approximately 20 kg.
o Perform the put out of operation procedure before moving the system.
u Put out of operation (p. 134)
o Do not attempt to relocate or transport the system without the assistance of
another individual.
o Do not attempt to lift the system with a Fluid Pack and AutoQC Pack in the
system. In addition, make sure that before lifting, there are no other objects
attached to the system.
o Lift the system only from its designated holding points.
u Holding points (p. 87)
Errors in installation Only trained Roche Service representatives may install the system.
o Leave installation that is not described to trained Roche Service representatives.
Disposal A biohazardous system may lead to infection.
o Treat the system as biohazardous waste. Decontamination (the combination of
processes including cleaning, disinfection, and/or sterilization) is required before
reuse, recycling, or disposal of the system.
o Dispose of the system according to the local regulations. For more information,
contact your Roche service representative.
Operating conditions
NOTICE
Unsuitable operating conditions
Operation outside of the specified ranges may lead to incorrect results or malfunction of
the system.
r Use the system indoors only, and avoid heat and humidity outside of the specified
range.
r Make sure that the system’s ventilation openings always remain unobstructed.
r Keep the analyzer away from all sources of liquids, such as sinks and wash basins.
r Keep the operating instructions undamaged and available for use. Operating
instructions have to be easily accessible for all users.
Electromagnetic interference Strong electromagnetic fields (originating from unshielded radio-frequency sources)
can interfere with proper operation and may lead to malfunction of the system and
incorrect results.
o Do not use this system near sources of strong electromagnetic fields because these
fields can interfere with its proper operation.
o Evaluate the electromagnetic environment before you operate the system.
o Take measures to mitigate the interference.

Roche Diagnostics
20 Instructions for Use · Version 13.0
1 Safety information cobas b 123 POC system
Safety precautions
Electrical safety
CAUTION
Electrical shock
Removing the covers of electronic equipment can cause electric shock because there are
high-voltage parts inside.
r Do not attempt to work in any electronic equipment.
r Do not remove any cover of the system except those covers specified in the
instructions.
r Only Roche Service representatives may install, service, and repair the system
Electrical specifications Comply with the following electrical specifications when operating the
cobas b 123 POC system:
o The power cable has to be connected into a grounded power socket. If you
connect the instrument to an extension cable, the extension cable has to be
connected into a grounded power socket.
o The power cable has to conform to country-specific requirements. Contact your
Roche Service representative for more information.
o Only use electrical accessories, such as power cables, provided by Roche. Do not
alter any electrical equipment on the instrument, or any electrical accessories that
accompany the system.
Biohazardous materials
Infectious samples Contact with samples containing material of human origin may result in infection.
All materials and mechanical components associated with samples containing
material of human origin are potentially biohazardous.
o Follow standard laboratory practices, especially when working with biohazardous
material.
o Keep all covers closed while the system is operating.
o Follow the instructions in this manual when performing procedures with the top
cover of the system removed.
o Wear appropriate personal protective equipment.
o If any biohazardous material is spilled, wipe it up immediately and apply a
disinfectant.
o If sample or waste comes into contact with your skin, wash the affected area
immediately with soap and water and apply a disinfectant. Consult a physician.
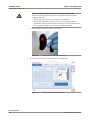
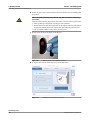
Sharp objects Contact with probes or needles may result in infection.
o When you wipe probes or needles, use several layers of gauze and wipe from the
top down.
o Take care not to puncture yourself.
o Wear appropriate personal protective equipment. Take extra care when working
with lab gloves, which can easily be pierced or cut, leading to infection.
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
Page is loading ...
-
 1
1
-
 2
2
-
 3
3
-
 4
4
-
 5
5
-
 6
6
-
 7
7
-
 8
8
-
 9
9
-
 10
10
-
 11
11
-
 12
12
-
 13
13
-
 14
14
-
 15
15
-
 16
16
-
 17
17
-
 18
18
-
 19
19
-
 20
20
-
 21
21
-
 22
22
-
 23
23
-
 24
24
-
 25
25
-
 26
26
-
 27
27
-
 28
28
-
 29
29
-
 30
30
-
 31
31
-
 32
32
-
 33
33
-
 34
34
-
 35
35
-
 36
36
-
 37
37
-
 38
38
-
 39
39
-
 40
40
-
 41
41
-
 42
42
-
 43
43
-
 44
44
-
 45
45
-
 46
46
-
 47
47
-
 48
48
-
 49
49
-
 50
50
-
 51
51
-
 52
52
-
 53
53
-
 54
54
-
 55
55
-
 56
56
-
 57
57
-
 58
58
-
 59
59
-
 60
60
-
 61
61
-
 62
62
-
 63
63
-
 64
64
-
 65
65
-
 66
66
-
 67
67
-
 68
68
-
 69
69
-
 70
70
-
 71
71
-
 72
72
-
 73
73
-
 74
74
-
 75
75
-
 76
76
-
 77
77
-
 78
78
-
 79
79
-
 80
80
-
 81
81
-
 82
82
-
 83
83
-
 84
84
-
 85
85
-
 86
86
-
 87
87
-
 88
88
-
 89
89
-
 90
90
-
 91
91
-
 92
92
-
 93
93
-
 94
94
-
 95
95
-
 96
96
-
 97
97
-
 98
98
-
 99
99
-
 100
100
-
 101
101
-
 102
102
-
 103
103
-
 104
104
-
 105
105
-
 106
106
-
 107
107
-
 108
108
-
 109
109
-
 110
110
-
 111
111
-
 112
112
-
 113
113
-
 114
114
-
 115
115
-
 116
116
-
 117
117
-
 118
118
-
 119
119
-
 120
120
-
 121
121
-
 122
122
-
 123
123
-
 124
124
-
 125
125
-
 126
126
-
 127
127
-
 128
128
-
 129
129
-
 130
130
-
 131
131
-
 132
132
-
 133
133
-
 134
134
-
 135
135
-
 136
136
-
 137
137
-
 138
138
-
 139
139
-
 140
140
-
 141
141
-
 142
142
-
 143
143
-
 144
144
-
 145
145
-
 146
146
-
 147
147
-
 148
148
-
 149
149
-
 150
150
-
 151
151
-
 152
152
-
 153
153
-
 154
154
-
 155
155
-
 156
156
-
 157
157
-
 158
158
-
 159
159
-
 160
160
-
 161
161
-
 162
162
-
 163
163
-
 164
164
-
 165
165
-
 166
166
-
 167
167
-
 168
168
-
 169
169
-
 170
170
-
 171
171
-
 172
172
-
 173
173
-
 174
174
-
 175
175
-
 176
176
-
 177
177
-
 178
178
-
 179
179
-
 180
180
-
 181
181
-
 182
182
-
 183
183
-
 184
184
-
 185
185
-
 186
186
-
 187
187
-
 188
188
-
 189
189
-
 190
190
-
 191
191
-
 192
192
-
 193
193
-
 194
194
-
 195
195
-
 196
196
-
 197
197
-
 198
198
-
 199
199
-
 200
200
-
 201
201
-
 202
202
-
 203
203
-
 204
204
-
 205
205
-
 206
206
-
 207
207
-
 208
208
-
 209
209
-
 210
210
-
 211
211
-
 212
212
-
 213
213
-
 214
214
-
 215
215
-
 216
216
-
 217
217
-
 218
218
-
 219
219
-
 220
220
-
 221
221
-
 222
222
-
 223
223
-
 224
224
-
 225
225
-
 226
226
-
 227
227
-
 228
228
-
 229
229
-
 230
230
-
 231
231
-
 232
232
-
 233
233
-
 234
234
-
 235
235
-
 236
236
-
 237
237
-
 238
238
-
 239
239
-
 240
240
-
 241
241
-
 242
242
-
 243
243
-
 244
244
-
 245
245
-
 246
246
-
 247
247
-
 248
248
-
 249
249
-
 250
250
-
 251
251
-
 252
252
-
 253
253
-
 254
254
-
 255
255
-
 256
256
-
 257
257
-
 258
258
-
 259
259
-
 260
260
-
 261
261
-
 262
262
-
 263
263
-
 264
264
-
 265
265
-
 266
266
-
 267
267
-
 268
268
-
 269
269
-
 270
270
-
 271
271
-
 272
272
-
 273
273
-
 274
274
-
 275
275
-
 276
276
-
 277
277
-
 278
278
-
 279
279
-
 280
280
-
 281
281
-
 282
282
-
 283
283
-
 284
284
-
 285
285
-
 286
286
-
 287
287
-
 288
288
-
 289
289
-
 290
290
-
 291
291
-
 292
292
-
 293
293
-
 294
294
-
 295
295
-
 296
296
-
 297
297
-
 298
298
-
 299
299
-
 300
300
-
 301
301
-
 302
302
-
 303
303
-
 304
304
-
 305
305
-
 306
306
-
 307
307
-
 308
308
-
 309
309
-
 310
310
-
 311
311
-
 312
312
-
 313
313
-
 314
314
-
 315
315
-
 316
316
-
 317
317
-
 318
318
-
 319
319
-
 320
320
-
 321
321
-
 322
322
-
 323
323
-
 324
324
-
 325
325
-
 326
326
-
 327
327
-
 328
328
-
 329
329
-
 330
330
-
 331
331
-
 332
332
-
 333
333
-
 334
334
-
 335
335
-
 336
336
-
 337
337
-
 338
338
-
 339
339
-
 340
340
-
 341
341
-
 342
342
-
 343
343
-
 344
344
-
 345
345
-
 346
346
-
 347
347
-
 348
348
-
 349
349
-
 350
350
-
 351
351
-
 352
352
-
 353
353
-
 354
354
Roche cobas b 123 <2> User manual
- Type
- User manual
- This manual is also suitable for
Ask a question and I''ll find the answer in the document
Finding information in a document is now easier with AI
Related papers
-
Roche cobas b 123 <2> User manual
-
Roche cobas b 123 <2> User manual
-
Roche cobas IT 1000 User manual
-
Roche cobas p 480 v2 User manual
-
Roche cobas b 221<4>=OMNI S4 system User manual
-
Roche cobas b 123 <2> User manual
-
Roche cobas c 702 User manual
-
Roche cobas IT 1000 User manual
-
Roche cobas c 702 User manual
-
Roche cobas c 702 User manual
Other documents
-
Tripp Lite Minicom Power-On Cable Quick start guide
-
Tripp Lite Minicom Power-On Cable Owner's manual
-
 palintest SA1100 User manual
palintest SA1100 User manual
-
Magellan LeadCare Ultra User manual
-
Magellan LeadCare II User manual
-
Abbott Afinion 2 User manual
-
PATH G6PD User guide
-
Korg microSAMPLER Easy Start Guide
-
Advantech POC-IPSM90CS3 User manual
-
 Tokyo Boeki Medisys BIOLIS 30I User's And Operator's Manual
Tokyo Boeki Medisys BIOLIS 30I User's And Operator's Manual



































































































































































































































































































































































